Focus: Cancer
Leading the Research for Cancer Cures
The cancer cells that don’t behave how you think
WHO: Yaara Oren, Zuckerman Faculty Scholar, Tel Aviv University
RESEARCH PROMISE:
In The Oren Lab, Dr. Oren and her team study persister cells to understand “why we haven’t cured cancer yet.” She explains that there are cells that should die, yet they persist – without demonstrating any changes or mutation to their genetic code – and clearly survive, likely driving a relapse.
“We are looking at alternative methods, such as epigenetics and post-translational modifications, rather than a mutation, and focusing on non-genetic traits that can be inherited from mother to daughter cell to tackle a question which has far-reaching clinical implications. These cells are one of the biggest problems, since most patients die of relapse, not from their original disease.”
The study of persister cells is a new field. There are currently a handful of labs in the world dedicated to going against the mainstream and studying these rare cells.
Listen to Dr. Oren describe this growing field and its significance for cancer research and treatment on The Global Connection.
The hidden communication systems that influence how tumors travel and how the immune system responds
WHO: Inbal Wortzel, Zuckerman Faculty Scholar, Tel Aviv University
RESEARCH PROMISE:
Dr. Wortzel’s research focuses on metastasis and the hidden communication systems that influence how tumors travel and how the immune system responds. Her work could lead to strengthening immune-based therapies to prevent cancer from spreading.
The Wortzel Lab investigates how cancer cells communicate with their environment through extracellular vesicles (EVs), focusing on DNA associated with EVs. Her research uncovers how EV-DNA shapes tumor–immune interactions, influences anti-tumor immunity, and affects metastatic progression. By combining advanced genomics, imaging, and in-vivo cancer models, Dr. Wortzel and her team aim to decode the molecular mechanisms governing EV-DNA biology and leverage this knowledge to improve early detection and develop new therapeutic strategies against metastasis.
Elucidating the hierarchy of high-order assembly formation in cancer, immunity, and neurobiology
WHO: Liron David, Zuckerman Faculty Scholar, Ben Gurion University of the Negev
RESEARCH PROMISE:
The Liron David Lab investigates essential proteins of the immune system which play central roles in inflammatory processes. The lab’s main interest is the hierarchy of high order assembly formation in cancer, immunity and neurobiology, a relatively new concept which states that only upstream molecules can trigger the assembly of downstream molecules. This leads to a tightly regulated “all-or-none” response in the cell, a characteristic for immune pathways, and provides protection against malignant cells.
The research conducted in the lab integrates structural biology, biochemistry, and cell biology to uncover the mechanisms that govern these proteins and their related pathways. By understanding these molecular interactions, the work forms a foundation for the development of targeted therapies and treatments, ultimately aiming to improve outcomes for patients affected by cancer and immune-related disorders.
Immune-stromal communication from basic science to translational applications
WHO: Ariella Glasner, Zuckerman Faculty Scholar, Technion -Israel Institute Of Technology
RESEARCH PROMISE:
Immune cells routinely detect and destroy cancer cells, but tumors have evolved ways to evade this defense. Modern immunotherapies target these evasion tactics, yet their effectiveness varies by patient and cancer type. Advancing treatment depends on a deeper understanding of immune-tumor dynamics.
The Glasner Lab maps interactions between immune cells and the tumor microenvironment (TME), focusing on regulatory programs, spatial effects, and genetic/epigenetic mediators. His team tests candidates for combination therapy using in-vivo models and tumor slice cultures from mice and humans to identify molecular mechanisms that may enhance immunotherapy outcomes.
Integrating genetic data and epidemiological insights to understand pre-cancerous conditions and develop models for personalized risk prediction—a truly innovative approach shaping the future of healthcare.
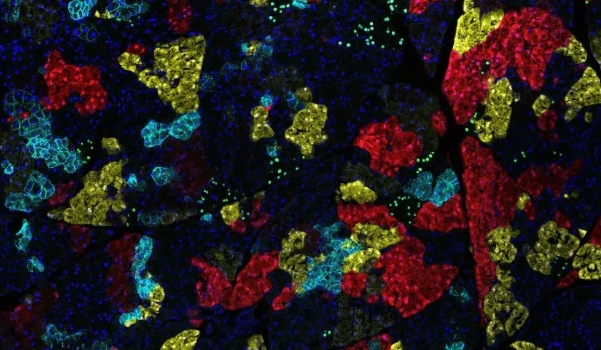
WHO: Geffen Kleinstein, Zuckerman Faculty Scholar, University Of Haifa
RESEARCH PROMISE:
Geffen Kleinstern’s lab focuses on analyzing genome-wide association studies (GWAS), tumor sequencing data, and epidemiological data, to explore the etiology and prognosis of cancer and pre-cancerous conditions, and develop inherited and tumor genetic scores for prediction models. These scores may be used for disease prevention and to improve clinical guidelines in disease management.
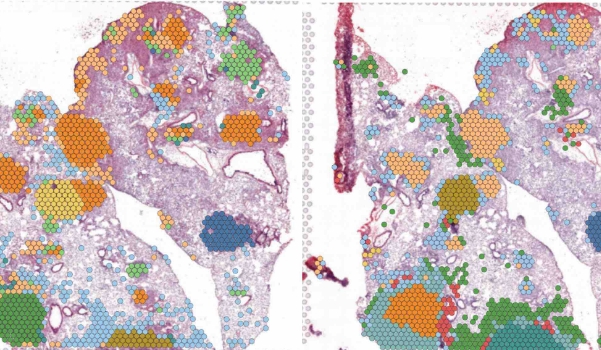
Advanced cellular mapping reveals why some cancer cells evade treatment while others succumb
WHO: Itay Tirosh, Zuckerman Faculty scholar at Weizmann Institute of Science
RESEARCH PROMISE:
Cracking the Code of Tumor Heterogeneity – Congratulations to Zuckerman Faculty Scholar Itay Tirosh of the Department of Molecular Cell Biology at Weizmann Institute of Science on the publication of “Single-cell analysis highlights the significance of malignant cell IFN/MHC-II for immunotherapy response in head and neck squamous cell carcinoma,” in Cell Reports Medicine. Using advanced RNA sequencing, Dr. Tirosh and his team identified specific cell subpopulations that drive invasion or resist standard treatments. Their research provides a vital blueprint for differentiation therapy, an approach aimed at rendering resistant cells harmless and refining the future of precision medicine.
